Table of contents
Continued from Part 1…
CY2017 Data Integrity Drug GMP Warning Letters and Trends from the Past Ten Years
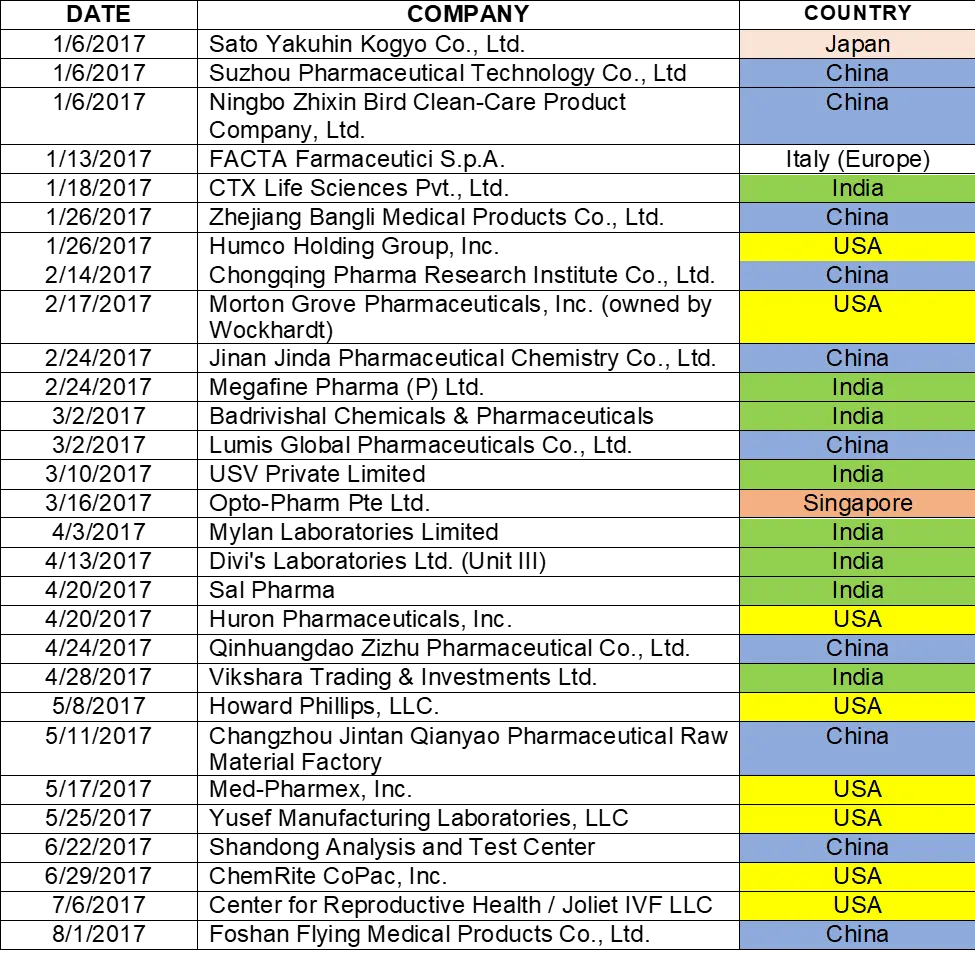
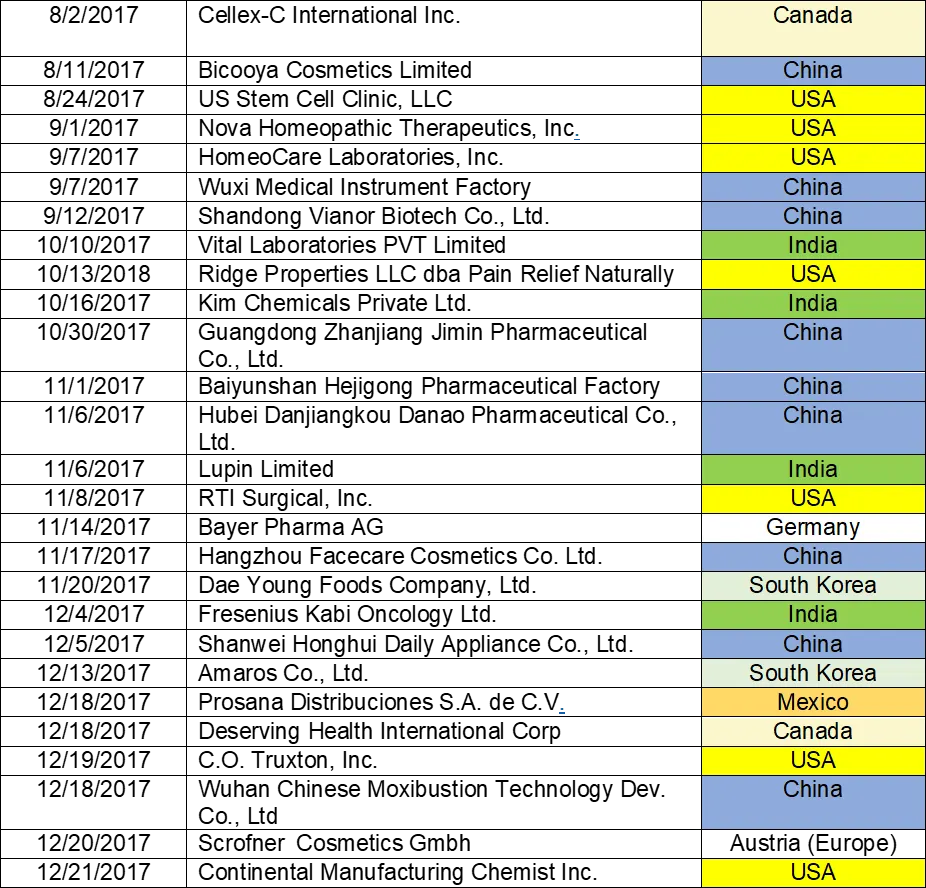
Table 1:
- Lists the warning letters that include data integrity deficiencies, the date of issuance, and the country location of the facility.
- The country column is color-coded, and I consolidate all European countries into a single group in subsequent tables and figures.
- FDA issued 82 warning letters, excluding those issued to compounding pharmacies and outsourcing facilities in CY2017.
- 56 warning letters included a data integrity component, a total of 68% of the warning letters.
TABLE 1: CY2017 Drug Warning Letters with Data Integrity Deficiencies
As mentioned in the background section, FDA began enforcement in this area nearly 20 years ago.
Table 2 and Figure 2:
- Present data over the last 10 years, CY2008 through CY2017.
- During this time, the number of warning letters including this topic ranged from 4 to 6 from 2008 through 2013, doubled in CY2014 to 10.
- The number of warning letters was followed by a marked increase between CY2015 through the current year, increasing from 15 in 2015 to 41 in 2016 and 56 in 2017.
- The number of countries associated with these warning letters also increased similarly, and in 2017 9 countries were associated with the sites that were subject to the warning letters.
Table 2: Number of Data Integrity Associated Warning Letters by Country CY2008 – 2017
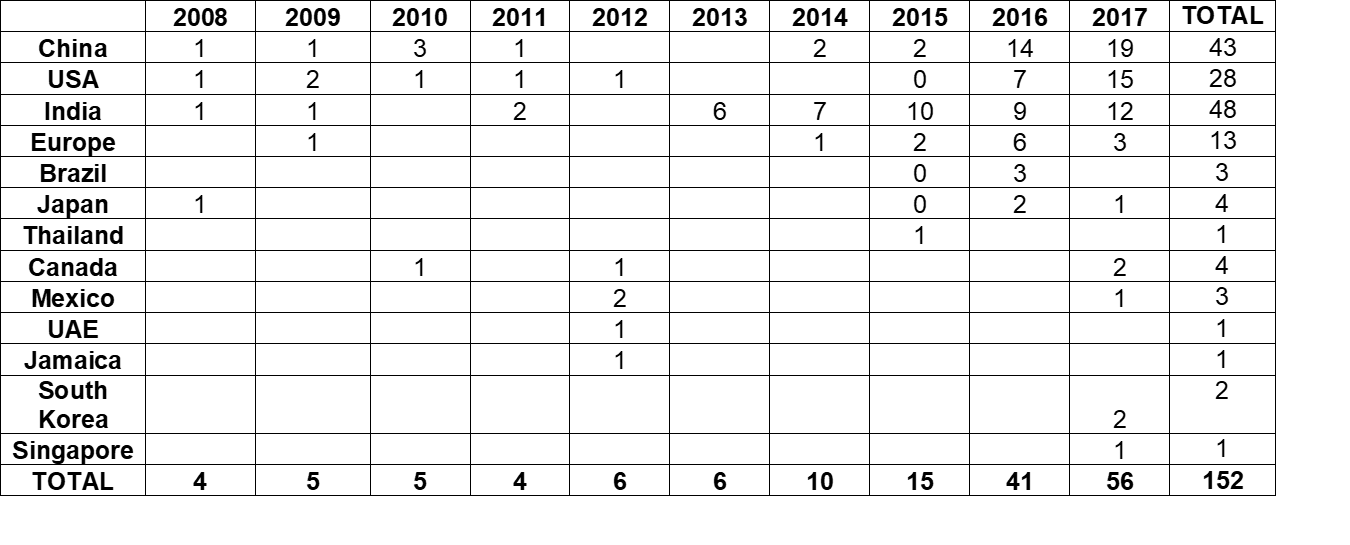
Figure 2: Data Integrity Associated Warning Letters, CY2008 – CY2017
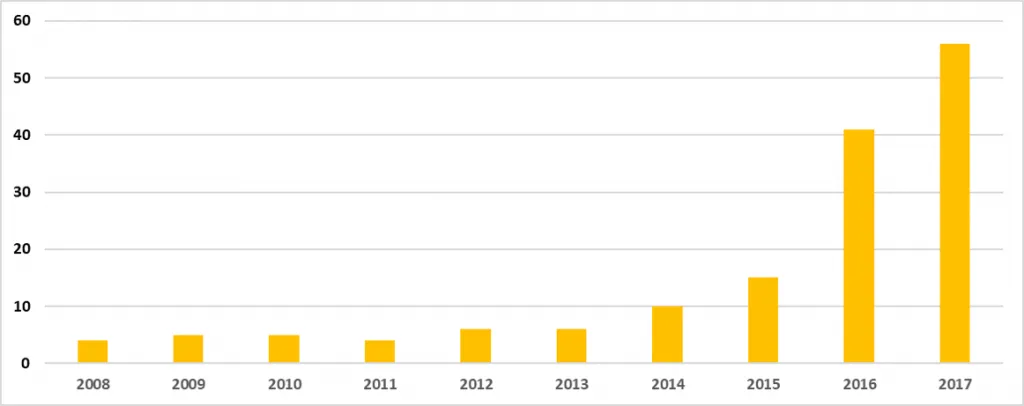
Table 4:
- Compares the number and percentage of warning letters citing data governance and data integrity in both the past 10 years and the most recent 3 years.
- In the past 10 years, sites in India have been the subject in the most warning letters of this type, whereas in the past 3 years, China rose to the head of the list.
- Overall, the US has received approximately 20% of the warning letters, European countries have received approximately 10%, and the rest of the world claim approximately 12%.
Table 4: Geographic Totals and Percentage, 2015 – 2017 and 2008- 2017
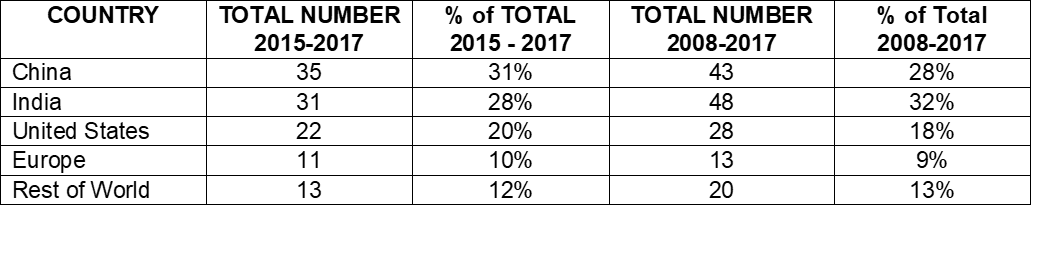
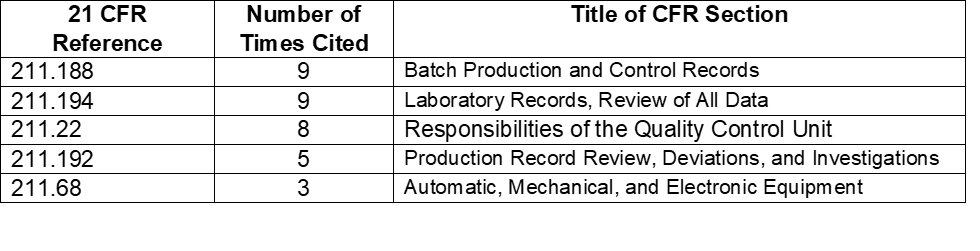
Table 5: Regulations Cited in 2017 Data Integrity Associated Drug Warning Letters
Follow along here for Part 3 and this series’ conclusions…
About the Author
Barbara W. Unger is Govzilla’s Quality Expert and Editor-in-Chief of GMP Regulatory Intelligence. She formed Unger Consulting, Inc. in December 2014 to provide GMP Quality consulting services to the pharmaceutical and biopharmaceutical industry. At Amgen, she led the segment of the Corporate GMP Audit group at Amgen focused on API manufacturers, Quality Systems and Computers. She developed, implemented and maintained the GMP Regulatory Intelligence program for 8 years at Amgen Inc. This included surveillance, analysis and communication of GMP related legislation, regulations, guidance and industry compliance enforcement trends. This was an essential service and tool within the Corporate Audit function.
Unger Consulting Inc. | www.ungerconsulting.net | 805.217.9360





