Table of contents
The FDA’s August 2025 inspection of a domestic CMO serves as a sharp reminder that non-sterile does not mean low risk. Microbial control lapses in non-sterile operations can draw regulatory attention similar to sterile facilities.
Across the nine observations listed in the 483, investigators found systemic weaknesses from uncharacterized microbial findings and inadequate disinfectant validation to unvalidated cleaning of laboratory glassware. Six of the nine observations related directly to contamination control, while a repeat observation from a 2023 inspection concerned the quality unit’s delayed complaint investigations, underscoring broader issues in quality oversight and CAPA effectiveness.
The agency’s expectations are clear in its 2021 draft guidance, “Microbiological Quality Considerations in Non-Sterile Drug Manufacturing: Draft Guidance for Industry.” It emphasizes that non-sterile drug makers must proactively manage microbial risks through sound environmental monitoring, cleaning validation, and contamination control strategies rather than reactive testing.
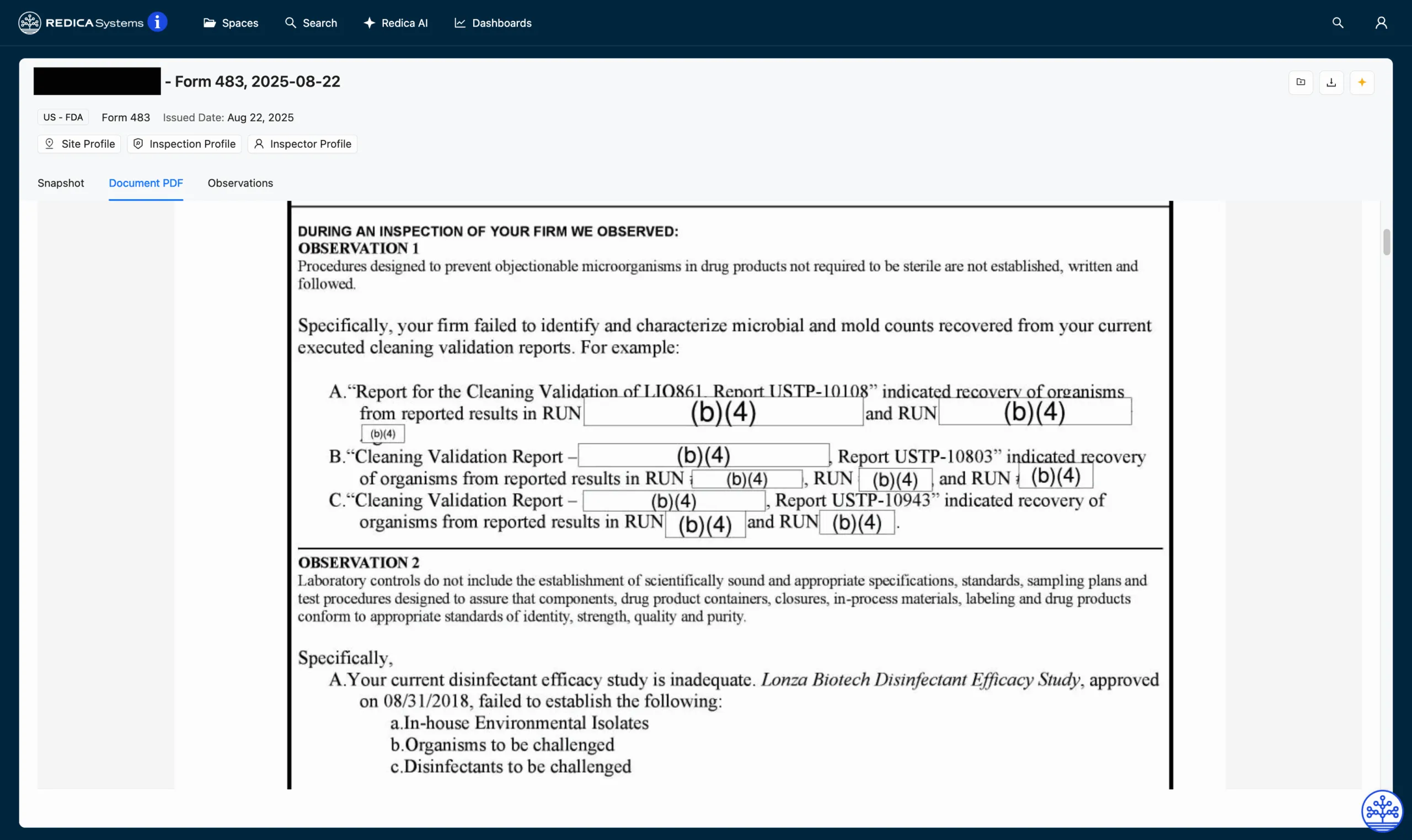
Figure 1 | Form 483 in the Redica Systems App
What stands out in this inspection is not just the technical noncompliance but the disconnect between procedures and practice. Validation data existed, but verification and follow-through did not. For quality executives, this reinforces that microbial control is as much about behavioral discipline and data integrity as it is about technical design.
The FDA’s message is consistent and clear: whether a product is sterile or not, contamination control and good documentation practices define a mature quality system. This 483 is a reminder to ensure your own teams aren’t treating “non-sterile” as “low risk.”
This inspection was a five-day, for-cause inspection that resulted in a seven-page, nine-observation 483 issued on August 22, 2025. The inspection was conducted by FDA investigators Kayla Sprague, Nicole Knowlton, and Santos Camara.
If you’re a current customer, you can view the full 483 directly in the Redica app. Not a customer yet? Fill out this form to access the 483.





