Table of contents
Do your suppliers and contract manufacturing organizations (CMOs) have robust GMP CAPA effectiveness? How can you ensure that your external partners adhere to manufacturing quality requirements?
[Related: Check out the top 483 observations for two of the leading CMOs for a three-year period. Download this FREE analysis here.]

During a session at the 2021 PDA/FDA Joint Regulatory Conference, Amgen Vice President of Corporate Quality Lisa Dierl covered how GMP CAPA effectiveness can serve as a competitive advantage, particularly when it comes to a company’s relationship with CMOs and suppliers.
CAPAs consist of actions to eliminate issues and prevent their recurrence (corrective action) and actions to prevent the issues from occurring again (preventive action).
As she put it, CAPA effectiveness “is a pay-me-now-or-pay-me-later scenario, and it is always less costly to do it right the first time.”
Dierl pointed out that multiple regulatory agencies require CAPAs.
“We all know CAPA is a legally required element of your quality management system (QMS) as well as your supplier’s QMS,” she said. “A big part of supplier management is the supplier CAPA management process and ensuring suppliers get to true root causes and eliminate defects and deviations.”
Performance Monitoring Critical for CAPA Effectiveness
Dierl explained that robust CAPA processes can enhance a company’s quality culture, reducing the costs associated with poor quality. These CAPA processes must also be followed by a company’s external suppliers and CMOs.
“An organization will be held accountable for the quality of the materials or services they are receiving from their suppliers as that quality affects the quality of the product they are delivering,” she explained. “Regulators view them as an extension of your business, so you should too.”
Performance monitoring, the measuring and tracking of incoming material inspection results, supplier deviations, and supplier CAPAs, is critical for ensuring quality at CMOs.
“It is really important to ensure suppliers provide appropriate notifications of changes and that a robust assessment is made of the potential consequences of these changes,” Dierl stressed. “Sometimes unintended consequences of seemingly small changes have a significant quality impact.”
Additionally, it is important to keep an open dialogue with suppliers and CMOs when it comes to CAPA alignment. This will help prevent issues that could potentially impact product supply later on.
Dynamite Caps Lead to Product Contamination
“Suppliers and CMOs need to align on initial impact risk assessments when a non-conformance (NC) or deviation is detected,” she said (Figure 1).
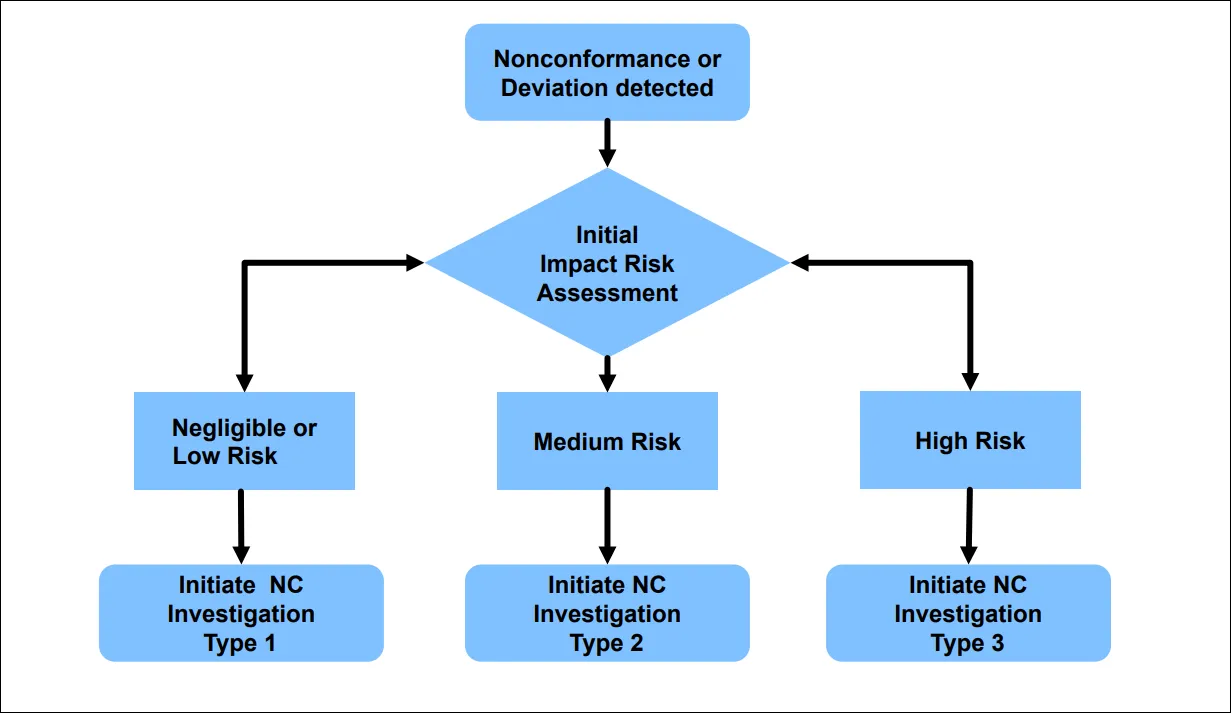
Detecting the non-conformance (a departure from specifications) or deviation is the first part of the process, followed by an initial impact risk assessment to understand potential risk to the product. Next, the risk is classified as low, medium, or high. The risk assessment then informs the type of investigation that follows.
“When speaking about effective CAPA practices, we all need to understand that the same applies whether it is an internal CAPA process or a supplier CAPA/corrective action report,” Dierl said.
She recounted a situation that occurred when she oversaw quality operations at a division of a major pharma company. Blue particles were found in a granulated product. Leadership assumed that an operator had accidentally dropped a ballpoint pen while performing formulation.
Taking a problem-solving approach, her team found that one of the ingredients in the product was calcium carbonate. The supplier quality lead visited the calcium carbonate supplier and during a walkthrough of their process noticed blue dynamite caps on the ground. An analysis of the caps showed the material matched the contaminant. Ultimately, holes in the metal filters at the supplier allowed material to pass through.
Sometimes unintended consequences of seemingly small changes have a significant quality impact
“This issue exemplifies the importance of using a defined process and not jumping to conclusions. We ended up solving for the appropriate root cause and never had the issue again,” she said.
Dierl recommends asking a series of what, where, when, and how questions to help identify the factors that contributed to an issue. She has also found that sometimes stakeholders do not understand the difference between a preventive or corrective action.
“To be clear, ‘preventive’ means ‘before it has happened.’ ‘Corrective’ means ‘after it has happened.’ Many times, I have people tell me, ‘it is a continuous improvement opportunity, so it is preventive.’ I always say, if a gap to a requirement exists, it is corrective, plain and simple. No positive spin can change these meanings, and this can be detrimental to the overall CAPA process.”
CAPA Issues a Common FDA 483 Observation
“A frequent 483 observation by regulatory agencies is that CAPAs were not actually implemented,” Dierl said. “You need to ensure, potentially, through audit, that these CAPAs have been appropriately implemented at your supplier or CMO or internally.”

Another crucial factor is ensuring that effectiveness verification can be performed.
“Effectiveness” refers to lack of root cause recurrence, and relates to causes, not symptoms, and is not merely a verification that actions were implemented.
“Effectiveness needs to be about whether the actions actually work. Throughout this process, monitoring or training effectiveness and human performance are foundational.”
A 4-Part Training Model
Dierl explained that when it comes to training, objective evidence is needed to support training programs and to avoid the “blame-and-retrain model.” [Author’s Note: For more on avoiding the “blame game” when it comes to training, read the article, “How GMP Leaders Can Turn Failure into Success: A Case Study.”]
She has employed a training model, both internally and externally, with some key suppliers and CMOs. This training model uses four different levels to evaluate training effectiveness:
- Level 1: At the end of training, trainee completes an evaluation form that asks about the training materials, trainer’s knowledge, and training environment
- Level 2: At the end of training, trainee completes an evaluation form that asks about the training materials, trainer’s knowledge, and training environment
- Level 3: Following training, the trainee must review an existing CAPA report, highlight the deviations, and then submit it for review
- Level 4: After completion of training, the training process is measured over a period of time by comparing a number of metrics (e.g., open CAPA investigations, CAPA investigation timeframe, recurring issues, etc.)
Human Error Another Important Part of CAPA Process
Additionally, identifying and preventing human errors that result in deviations is critical.
“90% of accidental losses begin with human error,” Dierl said. “Root causes of the vast majority of accidents are management system weaknesses. 80% of medical product recalls are due to incorrect expiration date or incorrect lot or batch number.”
She recounted how she handled an operator linked to three instances of formulation errors within a three-month period. Dierl observed the formulation process from afar, finding that the operator experienced frequent interruptions due to his proximity to other manufacturing activities.
“After observing this, one of our corrective actions was to put a red light in the area in which he was working. The operator’s work instructions included switching on that red light at the beginning of the process. Other operators were trained that when the red light was on, they were not to interrupt the operator.”
Throughout the next year, she continually evaluated the effectiveness of this corrective action. No further formulation mistakes occurred.
A Word About Quality Culture
When it comes to CAPA effectiveness, a strong quality culture is also critical and must extend to suppliers and CMOs.
“Quality does not own the supplier relationship, but when engaged effectively through leading effective supplier quality management practices, it improves the performance of the supply chain. Given the importance of the supply chain in today’s products, this creates benefits, not just in supplier quality, but across product management, manufacturing, and service as well,” she said.
Additional Resources
ORA Targets CAPA Effectiveness Check Deficiencies
Process Capability in Focus in FDA Device Inspections
Can a Robust Quality Culture Help Prevent GMP Issues Such as Data Integrity?
How GMP Leaders Can Turn Failure into Success: A Case Study