Table of contents
At the International GMP Conference held virtually in early March 2021 co-sponsored by the University of Georgia at Athens and FDA, FDA Office of Regulatory Affairs National Drug Expert Captain Ileana Barreto-Pettit presented FDA drug GMP warning letter trends and provided in-depth case studies from recent drug GMP inspections illustrating agency concerns and findings.
Barreto-Pettit has been with FDA for 22 years. She has been a drug investigator since 1999 and a Drug National Expert Investigator since 2017, and is a Captain in the U.S. Public Health Service Commissioned Corps.
[Related: For more from author Jerry Chapman, download our latest FREE report, “The Current Pharmaceutical Quality Landscape.”

The GMP inspection case studies Barreto-Pettit provided include an in-depth analysis of the findings, lessons learned, and how companies can avoid similar shortcomings. Areas examined in the case studies are:
- The first countrywide import alert issued by FDA
- Inadequate deviation investigation
- Inadequate product specifications and a product recall
- Process validation
- A different perspective on process validation and the culpability of the quality unit
Part 1 of this four-part series will cover Barreto-Pettit’s summary and analysis of recent FDA enforcement activities and the case study on a countrywide FDA import alert. The remaining case studies will be covered in the subsequent three parts.
Recent FDA Enforcement Action Statistics
Barreto-Pettit began her summary of recent FDA enforcement activities with a review of the agency’s inspection priorities during the last year and the impact of the COVID-19 pandemic on its activities.
Beginning in July 2020, onsite inspection activities resumed in domestic regions with lower COVID-19 risks based on data from the COVID-19 advisory rating system. “We conducted mission critical inspections domestically and in foreign countries throughout the pandemic,” Barreto-Pettit emphasized. She noted that “mission critical” inspections are those with the highest priority based on public health benefits or where the risks of not conducting the inspection outweigh the potential for investigators to potentially be exposed to COVID-19.
In reviewing the top FDA 483 observations for FY 2020, the FDA National Drug Expert commented that the same observations recur year after year, sometimes in a slightly different order (for a review of how and why 483 observations perennially repeat, see “Top 10 Pharma Inspection Findings from FDA, MHRA, and the Russian Drug Regulator”).She also reviewed the top ten 211 citations in FDA drug GMP warning letters for FY 2015 to FY 2020, through July 2020 (Figure 1).
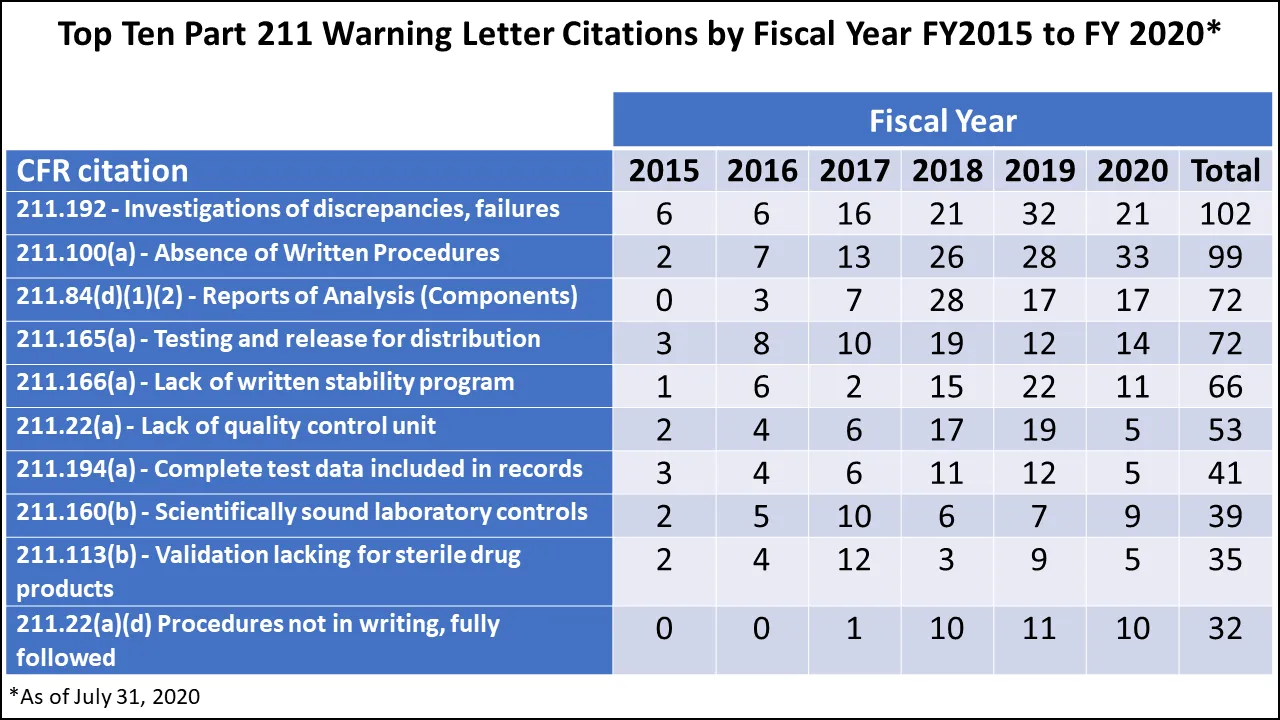
Barreto-Pettit commented, “As you can see, 21 CFR 211.100(a) for process validation was cited the most in warning letters in 2020. It was fourth in the 483s frequency but is the top citation in warning letters. And overall, over the years, inadequate investigations, which is 21 CFR 211.192, and process validation, have been the most common deficiencies that have been cited in warning letters.”
FDA FY2020 FDA Import Alerts
She next addressed import alerts. In addition to issuing warning letters, FDA has the authority to place non-compliant foreign firms under import alert. “This means that their products would not be allowed entry to the US while the import alert is in place. We use this regulatory tool to protect our consumers from potentially unsafe or ineffective products,” Barreto-Pettit explained. In 2020, the most common reasons for FDA issuing import alerts were equally distributed among inadequate GMPs, analytical test results, and refusal to inspect.
In 2020, Mexico received about 33.66% of the import alerts due to issues with analytical results for hand sanitizers, which contained methanol or did not meet the label content specifications. Import alerts for Europe were mostly for inadequate GMPs. In China, India, South Korea, and Taiwan, the main reason for import alerts was refusal to allow inspection. “If the manufacturer does not let us inspect the facility where they manufacture products that they bring into the United States, we will not allow their products to enter the U.S. market.”
Case Study: Hand Sanitizers With Deadly Toxins
The first case study the national drug expert discussed is from a warning letter issued to a hand sanitizer manufacturer in Mexico that imported its products into the United States. FDA collected samples and its test results showed that even though the drug product was labeled to contain 70% of the active ingredient as ethanol, it contained an average of 44% ethanol instead of 70% and the remaining was methanol, about 30%.
Therefore, these hand sanitizer drug products were adulterated under the Food, Drug and Cosmetic Act in that the active ingredient of ethanol was substituted in whole or in part with methanol.
“You may have seen this in the news,” Barreto-Pettit commented. She pointed out that methanol is not an acceptable ingredient for hand sanitizers and should not be used because it is toxic. When methanol is applied to the skin, it can cause dermatitis as well as more serious events facilitated by transdermal absorption resulting in systemic toxicity.
Substantial exposure to topical methanol can result in symptoms such as nausea, vomiting, headache, blurred vision, permanent blindness, seizures, coma, permanent damage to the nervous system or even death. Although all persons using these products on their hands are obviously at risk, young children who accidentally ingest these products and adolescents and adults who drink the product are at a potential life-threatening risk.
FDA has the authority to place non-compliant foreign firms under import alert
After confirming the test results, FDA issued a warning letter to the firm and an import alert to prevent its products from continuing to come into the United States. It also requested a recall of all the affected drug batches already distributed in the US. The warning letter was issued for an adulterated product, which means a product that was not manufactured under CGMPs or does not meet specifications.
Barreto-Pettit pointed out that the agency had not conducted an inspection of the facility. “We asked for a detailed investigation into how these hand sanitizer drug products manufactured at the facility and labeled as containing ethanol were substituted in part or in whole with methanol. We also requested a list of the raw materials used to manufacture all their hand sanitizer drug products, including the supplier’s names, addresses, and contact information.”
Import Alert Expanded to Include Entire Country
She explained that the requested information was useful to the agency because the manufacturer receiving the warning letter was not the only one having this kind of problem. FDA found many others that had methanol contaminated hand sanitizers and it needed to investigate whether the root cause of the problem was a supplier providing contaminated ethanol to multiple manufacturers.
FDA also requested a list of all the batches of any hand sanitizer drug products that were shipped to the United States by the firm and a full reconciliation of all the materials they distributed. “And since we had not done an inspection, we wanted to see how it was manufactured,” Barreto-Pettit said. “So, we requested copies of the complete batch records for all the batches that were distributed to the United States.”
In January 2021, FDA issued a MedWatch announcement and placed all alcohol-based hand sanitizers coming from Mexico on import alert to help stop products that are in violation from entering the United States until it could review the products’ safety. “And what we at FDA have seen is a significant number of hand sanitizer products from Mexico that were labeled to contain ethanol test positive for methanol or 1-propanol contamination.”
A subsequent FDA analysis of alcohol-based hand sanitizers imported from Mexico found that 84% of the samples analyzed by the agency from April to December of 2020 were not in compliance with FDA regulations or their specifications. More than half of the samples were found to contain toxic ingredients including methanol and/or 1-propanol at extremely dangerous levels.
Under this import alert, alcohol-based hand sanitizers from Mexico that are offered for import are subject to heightened FDA scrutiny. FDA import staff may detain the shipment. As part of their entry review, they will consider any specific evidence offered by the importers or the manufacturers that the hand sanitizers were manufactured according to US CGMPs. This is the first time the FDA has issued a country-wide import alert for any category of drug product.
[Editor’s Note: Part II covers a case study on an inadequate deviation investigation.]
[Related: Stay on top of the latest on data integrity, quality culture, and GMP employee development by downloading our latest FREE report here.]